Polyvinyl Alcohol
Polyvinyl Alcohol Product Information
Polyvinyl Alcohol is commonly used for adhesives, but it also widely used in industry and agriculture in emulsion polymerization, films and packaging, as well as oil field cementing, paper, textiles, ceramics, and seed coating. As a top polyvinyl alcohol supplier, we are continually developing new applications and products featuring polyvinyl alcohol.
Polyvinyl Alcohol is manufactured from vinyl acetate monomer (VAM) via a multistep process. VAM is polymerized into polyvinyl acetate and then converted to polyvinyl alcohol.
Grades
Various grades of Polyvinyl Alcohol offer more versatility than a single grade PVOH. The key to working with Polyvinyl Alcohol lies in finding the right grade to suit your needs.
Standard Grades
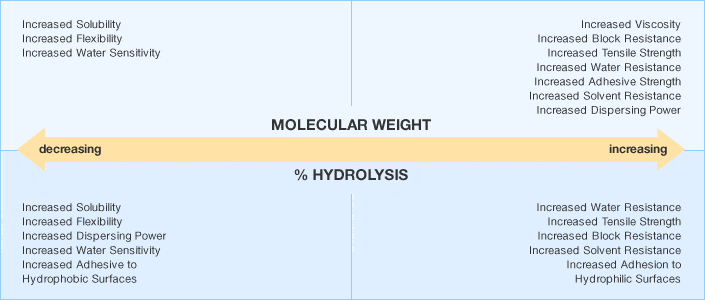
Individual Polyvinyl Alcohol grades vary in molecular weight and degree of hydrolysis. Molecular weight is generally expressed in terms of solution viscosity. The viscosities are classified as ultra low, low, medium and high, while degree of hydrolysis is commonly denoted as super, fully, intermediate and partially hydrolyzed. A wide range of standard grades are available.
- Polyvinyl Alcohols 805, 823 and 840 are improved versions of our standard polymerization grades (Polyvinyl Alcohol 205, 523 and 540 polyvinyl alcohols, respectively). These products offer a number of advantages in emulsion polymerization applications including improved water solubility and lower foaming.
- Polyvinyl Alcohol S-grades are similar to standard grades, but have a fine particle size so that 99+% of the product will pass through a U.S.S. 80-mesh screen. These grades are available commercially as Polyvinyl Alcohol 203S, 205S, 523S and 540S polyvinyl alcohols.
- Tackified Polyvinyl Alcohols are produced by controlled boration of super hydrolyzed and fully hydrolyzed grades. These borated alcohols yield viscous aqueous solutions, which have a tailored degree of tack and significantly reduce penetration when applied onto surfaces such as paper.
Polyvinyl Alcohol Solutions
Chemical Store is no longer offering pre-made solutions of some of PVOH grades.
Changes Occurring in the Properties of Polyvinyl Alcohol as the Degree of Hydrolysis and Molecular Weight Change
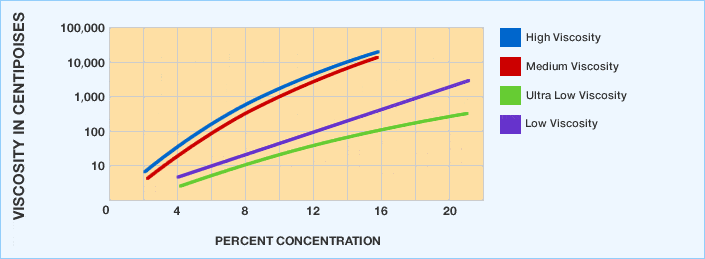
Viscosity / Concentration Relationships of Aqueous Polyvinyl Alcohol Solutions at 20 %
Polyvinyl Alcohol Molecular Weight
| Viscosity | Viscosity Type | Degree of Polymerization | Weight Average Molecular Weight Range |
|---|---|---|---|
| 3-4 cps | Ultra Low | 150-300 | 13,000-23,000 |
| 5-6 cps | Low | 350-650 | 31,000-50,000 |
| 22-30 cps | Medium | 1000-1500 | 85,000-124,000 |
| 45-72 cps | High | 1600-2200 | 146,000-186,000 |
Physical Properties
Polyvinyl Alcohol combines high tensile strength with ease of film formation. Additionally, Polyvinyl Alcohol resins show excellent adhesive and bonding characteristics. Partially hydrolyzed grades have better adhesion to hydrophobic surfaces.
The degree of hydrolysis affects the water sensitivity of both the resin and film. Water resistance increases with increasing hydrolysis. The super hydrolyzed grades should be used when maximum water resistance and humidity resistance are desired.
Polyvinyl Alcohol resins are generally unaffected by greases, petroleum hydrocarbons and animal or vegetable oils. Resistance to organic solvents increases with the degree of hydrolysis. Polyvinyl Alcohol film can be plasticized with glycerol or the lower molecular weight glycols. These materials generally act as humectants, holding water in the film.
Physical Properties of Polyvinyl Alcohol
| Appearance | White-to-cream granular powder |
| Bulk Density | 40 lbs/cu ft |
| Specific Gravity— of solid— of 10 wt % solid at 25°C | 1.27 – 1.311.02 |
| Thermal Stability | Gradual discoloration about 100°C; darkens rapidly above 150°C; rapid decomposition above 200°C |
| Thermal Conductivity, W/(m•K)3 | 0.2 |
| Electrical Resistivity, ohm•cm | (3.1 – 3.8) x 107 |
| Specific Heat, J/(g•K)b | 1.5 |
| Melting Point(unplasticized), °C | 230 for fully hydrolyzed grades; 180-190 for partially hydrolyzed grades |
| Tg, °C (dry film) | 75-85 |
| Storage Stability (solid) | Indefinite when protected from moisture |
| Flammability | Burns similarly to paper |
| Stability to Sunlight | Excellent |
Notes
a. To convert W/(m•K) to (Btu•in)/(h•ft2•F), divide by 0.1441.
b. To convert J to cal, divide by 4.184.
Chemical Reactions
Polyvinyl alcohol resins react in a manner similar to other secondary polyhydric alcohols. Esterification reactions of polyvinyl alcohol can be carried out with a number of compounds. A commercially important reaction is the formation of tackified PVOH using boric acid or borax to form cyclicesters. This reaction is very sensitive to pH, and an insoluble gel is formed above 4.5-5.0.
Other esterification reactions include those with chloroformate esters to yield polyvinyl carbonate, with urea to yield a polymeric carbamate ester, and with isocyanates to form substituted carbamate esters.
Another commercially important reaction is acetalization with aldehydes. Polyvinyl butyral is produced by the reaction of polyvinyl alcohol with butyraldehyde and is used in the production of the inner adhesive film for safety glass. Reaction with dialdehydes such as glyoxal or gluteraldehyde can be used to crosslink polyvinyl alcohol. Other reactions include ethoxylation, propoxylation and cyanoethylation.
Packaging and Storage
Polyvinyl Alcohol products are available in many packages:
- Granular/Powder product 25 KG, 500 KG, 1000 KG
- Other packaging may be available.